Official Newsjournal of the Illinois Council of Health-System Pharmacists

November 2021
Volume 47 Issue 4
Columns
March Board of Pharmacy Update
Collaborative Pharmaceutical Task Force Deliberations (March 2019)
Collaborative Pharmaceutical Task Force Deliberations (April 2019)
ICHP Leadership Spotlight Interview - Alifiya F. Hyderi, PharmD
ICHP Leadership Spotlight Interview - Jared Sheley, PharmD
Features
Student Chapter Video Contest 2019
Spring Meeting Platform Presentations
Spring Meeting Poster Abstracts
Spring Meeting Poster Abstracts
Spring Meeting Poster Abstracts
College Connection
Chicago State University College of Pharmacy
Midwestern University Chicago Collge of Pharmacy
Roosevelt University College of Pharmacy
Rosalind Franklin University College of Pharmacy
Southern Illinois University Edwardsville School of Pharmacy
University of Illinois at Chicago College of Pharmacy
More
ICHP Pharmacy Action Fund (PAC)
 Directly Speaking
Directly Speaking
Don't Just Network, Make Friends
by Scott A. Meyers, Executive Vice President

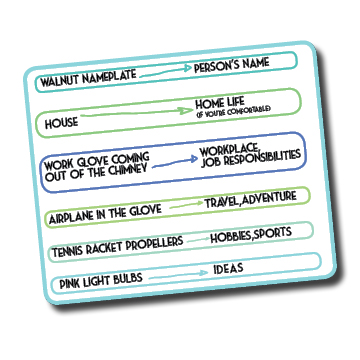
Now back to the real purpose of tying this to the networking event and the final part of this picture you’re creating in your mind. As the plane flies through the sky, it is surrounded by pink light bulbs instead of clouds. Why pink? I have no idea and didn’t write that in the notes but probably so you won’t forget them.
120x120.jpg) President's Message
President's Message
Spring Has Sprung
by Noelle Chapman, PharmD, BCPS, FASHP - ICHP President
 mily what was most important and make choices. Approximately a year and a half ago I had the distinct pleasure to hear Michelle Obama speak at the ASHP Midyear Clinical Meeting opening session. (Incidentally, I got to meet her afterwards thanks to my friend and past ICHP President, Todd Karpinski!) The entirety of her authentic, interview style keynote was inspirational to me, however, there was one portion that planted a seed in my mind. An audience member submitted a question asking Mrs. Obama if she ever planned to run for president. Her answer was a strong no for several reasons including family, privacy and politics, but she also said this (and I’m paraphrasing). Sometimes you need to vacate your seat at the table to give others a chance to grow. This concept of providing space for new ideas and opportunity and actively giving up something you earned made me look at my own choices in a different light.
mily what was most important and make choices. Approximately a year and a half ago I had the distinct pleasure to hear Michelle Obama speak at the ASHP Midyear Clinical Meeting opening session. (Incidentally, I got to meet her afterwards thanks to my friend and past ICHP President, Todd Karpinski!) The entirety of her authentic, interview style keynote was inspirational to me, however, there was one portion that planted a seed in my mind. An audience member submitted a question asking Mrs. Obama if she ever planned to run for president. Her answer was a strong no for several reasons including family, privacy and politics, but she also said this (and I’m paraphrasing). Sometimes you need to vacate your seat at the table to give others a chance to grow. This concept of providing space for new ideas and opportunity and actively giving up something you earned made me look at my own choices in a different light.Refresh and Renew
Columns
March Board of Pharmacy Update
Highlights of the March 2019 Meeting
by Scott A. Meyers, Executive Vice President
Collaborative Pharmaceutical Task Force Deliberations (March 2019)
We're Getting Closer But No Votes Yet
by Scott A. Meyers, Executive Vice President
· Patient counseling
· Drug regimen review
· Clinical conflict resolution
· Administration of immunizations
· Transfer of prescriptions
· Receiving of telephone orders for controlled
substances
Collaborative Pharmaceutical Task Force Deliberations (April 2019)
Still Closer to Voting, but Different Opinions Appear
by Scott A. Meyers, Executive Vice Presiden
- Advertising or soliciting that may jeopardize the health, safety, or welfare of the patient including but not limited to, advertising or soliciting that:
- Is false, fraudulent, deceptive, or misleading, or
- Makes any claim regarding a professional service or product or the cost or price thereof which cannot be substantiated by the licensee, or
- Requiring pharmacists to participate in such activities.
- Failure to provide a working environment for all pharmacy personnel that protects the health, safety, and welfare of a patient which includes, but is not limited to:
- Sufficient personnel to prevent fatigue, distraction, or other conditions that interfere with a pharmacist’s ability to practice with competency and safety or creates an environment that jeopardizes patient care
- Appropriate opportunities for uninterrupted rest periods and meal breaks.
- Adequate time for a pharmacist to complete professional duties and responsibilities including, but not limited to:
- Drug regimen review
- Immunizations
- Counseling
- Verification of the accuracy of a prescription; and
- All other duties and responsibilities of a pharmacist as specified in the Pharmacy Practice Act Administrative Rules Part 1330.
- Introducing external factors such as productivity or production quotas or other programs to the extent that they interfere with the ability to provide appropriate professional services to the public.
- Incenting or inducing the transfer of a prescription absent professional rationale.
- Anyone reporting violations of this section to the Department are specifically protected under the Illinois Whistle Blower Act (740 ILCS 174/15(b).
- Patient counseling
- Drug regimen review
- Clinical conflict resolution
Government Affairs Report
Legislative Day, New Lobbyists and Lots of New Bills
by Scott A. Meyers, Executive Vice President
 Following that, Liz Brown-Reeves and Mitch Schaben (busy in the Capitol at the time) were introduced as ICHP’s new legislative consulting team and lobbyists from LBR Consulting in Springfield. Liz and Mitch have hit the ground running and are working to keep you and our Government Affairs team informed.
Following that, Liz Brown-Reeves and Mitch Schaben (busy in the Capitol at the time) were introduced as ICHP’s new legislative consulting team and lobbyists from LBR Consulting in Springfield. Liz and Mitch have hit the ground running and are working to keep you and our Government Affairs team informed. I encourage you to review each one and look to see if your legislators are a sponsor of any of them. Below are the most important bills at this time:
ICHP Leadership Spotlight Interview - Alifiya F. Hyderi, PharmD
Leadership Profile

I am a Clinical Pharmacy Specialist and the PGY1 Pharmacy Residency Coordinator at RUSH University Medical Center in Chicago, Illinois. I work directly with the healthcare team to provide consistent patient care that ensures the appropriateness, effectiveness, and safety of patients’ medication use. I participate in daily team huddles that proactively address patients’ needs and problem solve how best to meet the needs of high-risk patients. In addition, I follow my passion of teaching and giving back to the profession by precepting pharmacy students and residents.
I am an advocate of patients actively participating in their own care. Early in my professional development, I realized that I can make a positive impact by empowering patients to make decisions and to instill a sense of control over their personal health. I once had an elderly patient who was unable to adhere to her medication regimen due to a combination of pill burden and the complexity of her regimen. I patiently went through the medication list and explained the indications for each of them in a way she could understand. We made a chart and organized medications that she can take in the morning, afternoon, and evening. I involved her in the decision-making process and addressed her questions and concerns. At the end of our conversation, I got a sense that she had a better understanding of her medication regimen. She also felt more confident in participating in her own self-care. A few months later, I met this same patient at the hospital cafeteria. I was surprised that she remembered me by my first name and was very appreciative of what I had done for her.
I was drawn to the pharmacy profession by seeing how accessible pharmacists are to people. When I was in high school, I remember going to a community pharmacy and being overwhelmed with the number of OTC products available. I went up to the pharmacy counter to consult the pharmacist. The pharmacist stepped out and walked with me to the aisle while asking me questions about my symptoms. Ultimately, he recommended a medication for my symptoms. He went a step beyond to explain how the product works, how I should take it, and what to do if my symptoms did not improve. In the years that followed, I learned more about the profession while shadowing pharmacists. I learned about the different opportunities that are available within the field and the ways pharmacists can provide care to patients. I also liked how pharmacy is a very respected profession.
When I moved to Illinois from California I wanted to get involved and network with pharmacists in the Illinois community. A few of my colleagues were active members of ICHP and seeing their involvement in ICHP got me interested in this organization.
I think ICHP is resourceful. The organization provides countless opportunities for pharmacy students, pharmacy technicians, and pharmacists. Through my involvement in ICHP, I have been able to personally benefit from the networking, leadership, and continuing education sessions. ICHP provides a platform to grow professionally, share ideas with fellow members, and realize that together we can do a lot to advance our profession.
Special thanks to my family for their unconditional love and support! I would also like to thank Dr. Tien Ng and Dr. Angela Rosenblatt for being excellent mentors to me throughout pharmacy school. I would also like to acknowledge Dr. Hina Patel for her endless support during my pharmacy residency, Dr. Nora Flint for being a positive influence in my career, and Dr. Kathryn Schultz, who has inspired me to expand my leadership influence within and outside of the institution where I work. I really admire each of their personalities and their commitment to the pharmacy profession.
What advice do you have for a student?
Don’t hesitate to step out of your comfort zone.
I enjoy working on DIY projects
I love Mediterranean and Indian cuisines, although I also love trying different cuisines.
Determined, diligent, empathetic. ■
ICHP Leadership Spotlight Interview - Jared Sheley, PharmD
Leadership Profile

I serve as the President of the Metro East Affiliate of ICHP (Metro East Society of Health System Pharmacists or “MESHP”), where I am responsible for planning and carrying out local affiliate meetings and working directly with the ICHP Board on relevant activities. I re-initiated the MESHP as an active chapter in 2015 and have served as President since that time. I am also the Director-elect of the Southern Region, where I will represent Southern Illinois members on the ICHP Board of Directors.
I am a Clinical Assistant Professor at the Southern Illinois University Edwardsville School of Pharmacy, teaching in a variety of clinical courses. I am also a Clinical Pharmacy Specialist (Internal Medicine) at HSHS St. Elizabeth’s Hospital, where I round with the inpatient teaching service with the Saint Louis University Family Medicine Residency Program. I also regularly contribute to educational activities for the Family Medicine Residency program, as well as precept APPE students, pharmacy residents, and medical residents.
I decided to go into the pharmacy profession as a high school senior after contemplating several healthcare related professions and shadowing pharmacists in multiple practice settings. I was excited about the knowledge pharmacists had regarding how specific medications worked and how they contributed to enhancing patient care. Every time I make a significant clinical intervention at work, or instill important knowledge and skills in pharmacists-in-training or physicians that I know will have a positive impact on multiple patients in the future, I know I have selected the right career.
On several occasions, I have identified areas for improvement and subsequently developed and delivered formal education to pharmacists, physicians, and nursing staff to impact positive changes in patient care.
Why do you stay involved in ICHP?
ICHP is great because of the many amazing people who dedicate their time to continually improving this already great organization!
I first joined ICHP as a student. After returning to Illinois following 2 years of residency training, I was encouraged by a few colleagues at SIUE to get more involved within ICHP and consider rejuvenating a local presence in the Metro East.
Special thanks to everyone who contributed to my professional growth and for making me who I am today in my career. I don’t have one person to point out, but I have gained something from many, many people along the way – so thank you to ALL of you! Also to my wife for all of her support throughout my school, post-graduate training, and busy work life!
Keep an open mind about your future and do not be afraid to change paths as you encounter new knowledge, experiences, or opportunities. Try to find a way to grow through every single experience.
Spending time with family (especially with my recent addition of twin boys) and friends, being outdoors and active (hiking/camping/kayaking/recreational sports), and becoming a self-taught handyman.
I was a state champion wrestler. ■
New Practitioners Network
Mentors Matter: Finding the Match to Make the Right Move
by Dalila Masic, PharmD PGY2 Critical Care Resident Loyola University Medical Center, Maywood, IL; Samantha Siepak, PharmD Candidate 2020 Midwestern University Chicago College of Pharmacy Downers Grove, IL; Megan Park Corsi, PharmD, MBA, DPLA Clinical Research Pharmacist The University of Chicago Medicine Chicago, IL
Professional Affairs
What are Position Statements and why does ICHP have them?
by Mary Lee, Director of Division of Organizational Affairs; Karin Terry, Director of Division of Professional Affairs
- When a new patient-specific challenge arises for which your facility does not have a policy or position.
- When updating your facility-specific policies.
- As a reference when preparing a presentation, in-service, etc.
- As a reference when evaluating a specific service or practice at your facility.
Professional Affairs
Call for Best Practice Award
The objective of the program is to encourage the development of new, innovative pharmacy practice programs or innovative approaches to existing pharmacy practice challenges in health systems within the state of Illinois.
- Innovativeness / originality
- Contribution to improving patient care
- Contribution to institution and pharmacy practice
- Scope of project
- Quality of submission
Each entry for the Best Practice Award must include a manuscript prepared as a Word document, double-spaced using Times New Roman 12-pitch type. A header with the paper title and page number should appear on each page.The manuscript should not exceed 2000 words in length (not counting references), plus no more than a total of 6 supplemental graphics (tables, graphs, pictures, etc.) that are relevant to the program. Each picture, graph, figure, and table should be mentioned in the text and prepared as a separate document clearly labeled
- Introduction, Purpose, and Goals of the program
- Description of the program
- Experience with and outcomes of the program
- Discussion of innovative aspects of programs and achievement of goals
- Conclusion
Submissions will only be accepted via online submission form. The manuscript will be forwarded to a pre-defined set of reviewers. Please do not include the names of the authors or affiliations in the manuscript to preserve anonymity.
- The program will be featured at the ICHP Annual Meeting. You will need to prepare a poster to present your program and/or give a verbal presentation. Guidelines will be sent to the winner.
- You may be asked to electronically submit your manuscript to the ICHP KeePosted™ for publishing as a continuing pharmacy education home study program.
- You will receive a complimentary registration to the ICHP Annual Meeting, recognition at the meeting and a monetary award distributed to your institution.
Email Trish Wegner at TrishW@ichpnet.org or go to www.ichpnet.org/pharmacy_practice/professional_practice/best_practices/ ■
Educational Affairs
Implementing Anticoagulation Dosing Guidance and Drug-Drug Interaction Procedures
by By Cody C. Anderson, PharmD Long Term Care Consultant Pharmacist, Omnicare; Megan Grischeau, PharmD, BCACP VISN12 Clinical Pharmacist, Anticoagulation Hub – Midwest Region 2016-2019 Captain James A. Lovell Health Care Center PGY2 Ambulatory Care Residency Program Director Veterans Health Administration Integrated Service Network (VISN12)
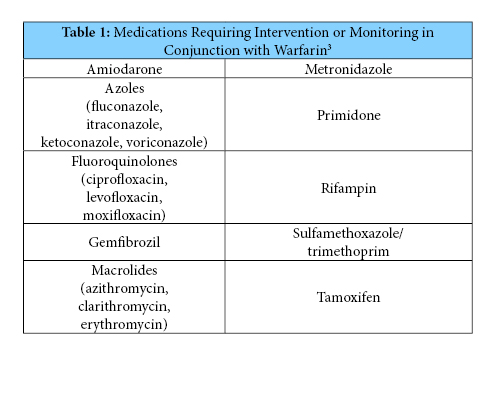
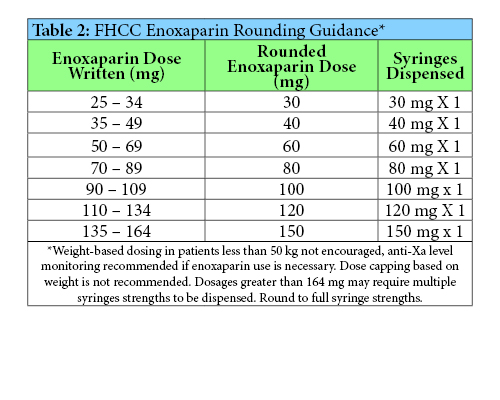
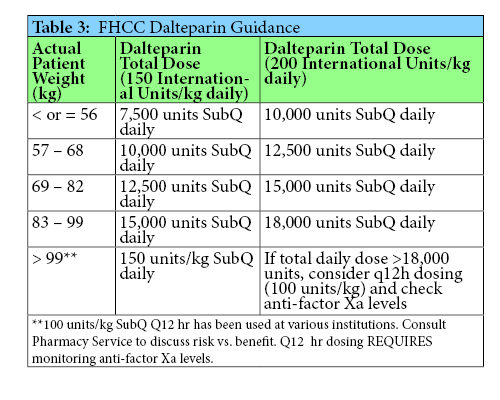
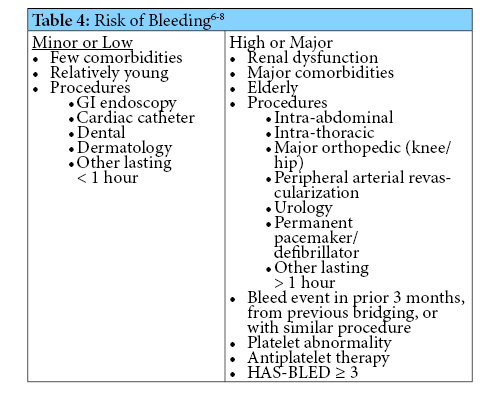
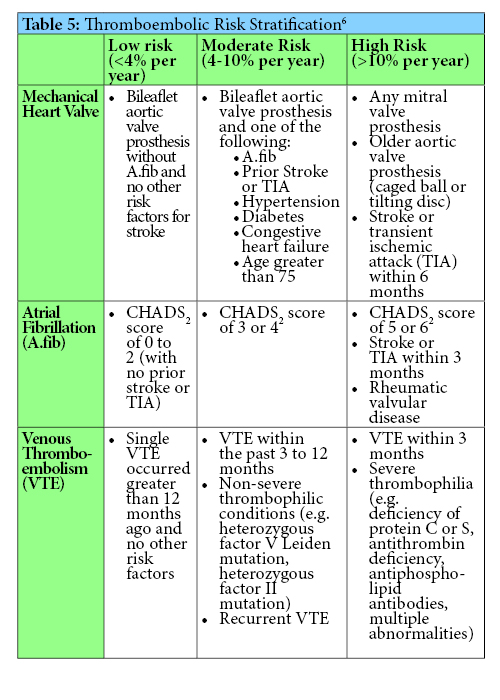
Since these processes were recently adopted, it is too soon to know their full impact. It can be difficult to change previous practice patterns, thus feedback from pharmacists and providers will be welcomed to maximize the efficacy and efficiency of the guidance and SOP. Time-in-therapeutic-range reports can be accessed in the VA data warehouse to potentially assess safety and efficacy of the process. However it is possible that some patients may not have an INR for four weeks following the drug interaction. At that time, the interacting drug may already be eliminated from the patient’s system and INR could have already returned to normal. Therefore, evaluation of the process may require generating reports of the above drug-drug interactions to assess if there was corresponding documentation in the patient’s medical record. Reassessment of the warfarin drug-drug interaction SOP may require re-education of pharmacy staff depending on activity of the CPS email group regarding interactions.
Young S, Bishop L, Twells L, et al. Comparison of pharmacist managed anticoagulation with usual medical care in a family medicine clinic. BMC Family Practice 2011;12:88.
Zhou S, Sheng X, Xiang Q, et al. Comparing the effectiveness of pharmacist-managed warfarin anticoagulation with other models: a systematic review and meta-analysis. J Clin Pharm Ther 2016;41:602-11.
Bungard T, Yakiwchuk E, Foisy M, et al. Drug interactions involving warfarin: practice tool and practical management tips. Can Pharmaceut J 2011;144(1):21-25.
Lovenox® [package insert]. Bridgewater, NJ: Sanofi-Aventis; October 2017.
Fragmin® [package insert]. New York, NY: Pfizer Inc; June 2017.
Douketis J, Spyropoulos A, Spencer F, et al. The perioperative management of antithrombotic therapy: American College of Chest Physicians evidence-based clinical practice guidelines (9th Edition). Chest 2012;141:326S-350S.
Lip GY, Frison L, Halperin J, et al. Comparative validation of a novel risk score for predicting bleeding risk in anticoagulated patients with atrial fibrillation. J Am Coll Cardiol 2011 Jan 11;57(2):173-80.
Doherty J, Gluckman T, Hucker W, et al. 2017 ACC expert consensus decision pathway for periprocedural management of anticoagulation in patients with non valvular atrial fibrillation. J Am Coll Cardiol 2017 Feb 21;69(7):871-898.
Duhl A, Paidas M, Ural S, et al. Antithrombotic therapy and pregnancy: consensus report and recommendations for prevention and treatment of venous thromboembolism and adverse pregnancy outcomes. AJOG Nov 2007;457:e1-57.e21.
Features
Looking For Some Leaders
How would you like to run for an ICHP Office?
by Scott A. Meyers, Executive Vice President
- President-Elect
- Secretary-Elect
- Director-Elect of the Division of Educational Affairs
- Director-Elect of the Division of Marketing Affairs
- Technician Representative-Elect
- NPN Chair-Elect
Legislative Day Highlights
Under the Dome #13 is Under our Belts
by Scott A. Meyers, Executive Vice President
.jpg) The 13th Annual Illinois Pharmacy Legislative Day - “Under the Dome” is the well-known tagline - was a rousing success on March 13th! More than 400 attendees trekked to Springfield to meet with legislators and this year, rally in the Capitol Rotunda as Representative Greg Harris, D-Chicago and Senator Andy Manar, D-Springfield unveiled two identical bills to regulate PBMs (Pharmacy Benefit Managers)! On the Monday and Tuesday prior to Leg Day, both legislators introduced amendments to HB0465 and SB0652 that would begin to provide pharmacies and patients with significant protections from the predatory practices of PBMs! During the course of the day, more than 20 legislators were convinced to sign on as co-sponsors to one of these two pieces of legislation!
The 13th Annual Illinois Pharmacy Legislative Day - “Under the Dome” is the well-known tagline - was a rousing success on March 13th! More than 400 attendees trekked to Springfield to meet with legislators and this year, rally in the Capitol Rotunda as Representative Greg Harris, D-Chicago and Senator Andy Manar, D-Springfield unveiled two identical bills to regulate PBMs (Pharmacy Benefit Managers)! On the Monday and Tuesday prior to Leg Day, both legislators introduced amendments to HB0465 and SB0652 that would begin to provide pharmacies and patients with significant protections from the predatory practices of PBMs! During the course of the day, more than 20 legislators were convinced to sign on as co-sponsors to one of these two pieces of legislation!
Student Chapter Video Contest 2019

Champions Subcommittee
Be a Champion!
by Julie Downen, Chairperson of Champions Subcommittee
ICHP/MSHP Spring Meeting
Poster Session Abstracts

Educational Affairs
2019 ICHP Spring Meeting Poster Abstracts
Please note: All research was to have results and
conclusions by the time of presentation. These may not be reflected in the
posted abstracts below.
PLATFORM PRESENTATIONS (Presented on Saturday, March 30, 2019)
Assessment of the Illinois Pharmacist’s Understanding of Palliative Care – Winner Best Platform Presentation
Exploration of Aztreonam in Combination with Avibactam or Vaborbactam for the Potential Treatment of Levofloxacin and/or Sulfamethoxazole-trimethoprim Resistant Strains of Stenotrophomonas Maltophilia Infections
********************************************************************************************************************
ORIGINAL RESEARCH POSTERS
On Site #1 - Operationalizing dispensing free naloxone out of a Level 1 Trauma Center Emergency Department (ED)
On Site #2 - Implementation of an enhanced recovery after surgery (ERAS) protocol in bariatric surgical patients to decrease post-operative nausea and vomiting: a retrospective chart review
On Site #3 - Impact of pharmacy process improvements within the emergency department on time to antibiotic administration and outcomes in patients with sepsis
On Site #38 - Value of pharmacy technician clinical support role in primary care
On Site #39 - Cost Savings Associated with a Therapeutic Conversion of Insulin Aspart to Insulin Lispro – Winner Best Original Poster
********************************************************************************************************************
ENCORE POSTERS
On Site #4 - The Financial Impact of Pharmacist Interventions in an Ambulatory Care Oncology Clinic Encore ICHP
On Site #5 - Student Pharmacist Driven Medication History Training Pilot Program Encore ICHP
On Site #6 - Assessing current gaps in practice and improving hospital adherence to ISMP’s Best Practice recommendations surrounding oral methotrexate use in non-cancer indications
On Site #7 - Impact of Team-Based Learning on Pharmacy Students’ Knowledge and Confidence in Searching Primary Literature Encore ICHP
On Site #40 - Comparing the efficacy of serum vancomycin concentrations of pharmacist-driven versus infectious disease physician-driven dosing of vancomycin – Winner Best Encore Poster
********************************************************************************************************************
STUDENT RESEARCH POSTERS
On Site #8 - Concurrent use of benzodiazepines and opioids: a drug utilization review based investigation in a community hospital
On Site #9 - A Retrospective, Single Center Study Evaluating COPD Management and Hospital Readmissions
On Site #10 - Does the Infusion Time of IVIG Matter in Kawasaki Disease?
On Site #11 – A comparison of renal outcomes among targeted immunotherapies – Canceled (Results and Conclusions were not available by the time of presentation)
On Site #12 - Analyzing the Role of Pharmacists in Overcoming Inadequate Documentation of Angiotensin Converting Enzyme Inhibitor Induced Drug Reactions in the Electronic Medical Record (Updated title)
On Site #41 - Nalbuphine as a Primary Parenteral Opioid for Acute Pain in a Large Community Hospital: A Retrospective Review.
On Site #42 - Dispense this, not that! Requirements for confident opioid dispensing
On Site #43 – A Retrospective, Single Center Study Evaluating Readmission Rates and Medications for Patients with Heart Failure with Preserved Ejection Fraction – Winner Best Student Poster
On Site #44 - Acute pain management for patients with an opioid dependence disorder receiving Suboxone, naltrexone or methadone
On Site #45 - Application of the Utah Bleeding Risk Score to LVAD Supported Patients at Advocate Christ Medical Center: A Retrospective Study (Updated title)
On Site #46 - Rocuronium versus succinylcholine in the traumatically injured brain: A prospective observational cohort (RVSTIB)
On Site #47 - Diluted single-syringe administration of adenosine for the rapid conversion of supraventricular tachycardia in the Emergency Department
********************************************************************************************************************
********************************************************************************************************************
********************************************************************************************************************
Spring Meeting Platform Presentations
Platform Presentation Abstracts
PLATFORM PRESENTATION ABSTRACTS
Platform Presentation – Student Research
Title: Assessment of the Illinois Pharmacist’s Understanding of Palliative Care – Winner Best Platform Presentation
Submitting Author: Blake Cornwell, PharmD Candidate
Authors: Blake Loran Cornwell, PharmD candidate, Southern Illinois University at Edwardsville School of Pharmacy; Katie Ronald, PharmD, BCPS, Professor, Pharmacy Practice, Southern Illinois University at Edwardsville School of Pharmacy.
Organization: Southern Illinois University at Edwardsville School of Pharmacy
Abstract
Purpose: The purpose of this palliative care survey was to assess Illinois pharmacists’ level of involvement and understanding related to palliative care.
Methods: The 22 question palliative care survey was distributed through Qualtrics after the IRB was approved. Once the survey was finalized an email was sent to the Illinois Pharmacists Association (IPhA) and the Illinois Council of Health-System Pharmacists (ICHP). These organizations then distributed the survey to their respective members. The email gave Illinois pharmacists an idea of what the research would be about and how it could benefit the pharmaceutical profession as a whole. Participants were able to access the survey through an anonymous link within the email. The survey was open to access from October 29, 2018 to November 30, 2018.
Results: A total of 152 responses were recorded with 131 of those participants completing the entire survey. The majority of pharmacists stated that they rarely (26.1%) or never (45.5%) engage in palliative care at their practice site. The majority of survey respondents selected that a goal of palliative care is improving quality of life (51.7%) and/or optimizing symptom management (43.6%) as opposed to halting the progress of life (2.6%) and/or doing whatever is necessary to keep patient alive (2.1%). When asked to rank objectives of palliative care respondents thought providing relief from pain and other distressing symptoms (30.9%) and enhancing patient’s quality of life (32.4%) were the most important palliative care objectives. When asked what challenge would make it the most difficult to educate patients about palliative care the 3 answers that made up the majority of responses were not enough time to discuss palliative care topics (37.3%), patients being unwilling to talk about palliative care topics (23.9%), and pharmacists’ lack of knowledge about palliative care topics (23.1%). Pharmacists were very interested in receiving additional education about pain management based on disease state (60.9%), symptom management based on disease state (53%), and mental health management for patient and family (45.1%). In addition to the topics listed above, most pharmacists were at least very interested or moderately interested in receiving additional education about communication with patient and family, spiritual needs, cultural beliefs re: death and dying, and legal aspects of patient care decisions.
Conclusions: The Illinois pharmacists who completed this survey displayed an understanding of the goals of palliative care and they believed enhancing quality of life as well as providing symptomatic relief was of the utmost importance for patients receiving palliative care. Despite multiple challenges making it difficult to educate patients about palliative care, pharmacists were willing to receive education about a plethora of palliative care topics to assist them in educating patients in the future. As the pharmacy profession progresses, the hope is that pharmacists will feel more comfortable discussing palliative care topics with patients and implementing palliative care management plans with other healthcare providers.
********************************************************************************************************************
Platform Presentation – Student Research
Title: Exploration of Aztreonam in Combination with Avibactam or Vaborbactam for the Potential Treatment of Levofloxacin and/or Sulfamethoxazole-trimethoprim Resistant Strains of Stenotrophomonas Maltophilia Infections
Submitting Author: Denise Lamm, PharmD Candidate
Authors: Denise Lamm, PharmD Candidate, Graduate Research Assistant, UIC College of Pharmacy; Mark Biagi, PharmD, Infectious Diseases Pharmacotherapy Fellow, UIC College of Pharmacy; Tiffany Wu, PharmD Candidate, Graduate Research Assistant, UIC College of Pharmacy; Kevin Meyer, PharmD Candidate, Graduate Research Assistant, UIC College of Pharmacy; Eric Wenzler, PharmD, Assistant Professor of Pharmacy Practice, UIC College of Pharmacy.
Organization: University of Illinois at Chicago College of Pharmacy
Abstract
Purpose: Given the widespread antibiotic resistance among Stenotrophomonas maltophilia and potential for toxicities and/or drug interactions to currently preferred agents, alternative therapies are needed. Aztreonam is the only β-lactam capable of avoiding hydrolysis by the L1 metallo-β-lactamase intrinsically expressed by S. maltophilia. Aztreonam remains susceptible to hydrolysis, however, by the L2 serine β-lactamase, thus making it ineffective for treating S. maltophilia. Combining aztreonam with a β-lactamase inhibitor with activity against L2 may restore aztreonam’s activity against S. maltophilia.
Methods: Thirty-seven S. maltophilia isolates resistant to levofloxacin and/or sulfamethoxazole-trimethoprim underwent MIC testing in triplicate by broth microdilution method according to CLSI guidelines. Modal MICs are reported and susceptibility interpretations were determined based on CLSI breakpoints for both levofloxacin and sulfamethoxazole-trimethoprim against S. maltophilia and Pseudomonas aeruginosa for aztreonam-based regimens. Time kill analyses for five isolates at standard inoculum (106) were performed in triplicate for aztreonam, aztreonam/avibactam, and aztreonam/vaborbactam. Time kills were performed at either fCmax or ¼, ½, 1, 2, and 4x the MIC. A >3 log10 reduction in CFU/mL from the starting inoculum was considered to be bactericidal. Synergy was considered to be ≥2 log10 reduction in CFU/mL compared to the most active agent alone.
Results: Only one of the 37 isolates (2.7%) was susceptible to aztreonam alone (MIC50/90> 128/>128 mg/L; MIC range 8->128 mg/L). Combining aztreonam with avibactam restored aztreonam susceptibility in 97.2% (35/36) of aztreonam-resistant isolates (MIC50/90 2/4 mg/L; MIC range 0.5-16 mg/L). Combining aztreonam with vaborbactam restored aztreonam susceptibility in 11.1% (4/36) of aztreonam-resistant isolates (MIC50/90 64/>128 mg/L; MIC range 2->128 mg/L). In time kill analyses, aztreonam alone failed to demonstrate bactericidal activity against any of the tested isolates at fCmax. Aztreonam/avibactam showed bactericidal activity at 4x MIC in 3/5 isolates while aztreonam/vaborbactam exhibited bactericidal activity against only 1/5 isolates at fCmax.
Conclusions: Combining aztreonam with avibactam resulted in restored susceptibilities at a higher rate than combing aztreonam with vaborbactam. This suggests that this combination could be an effective treatment alternative for S. maltophilia resistant to currently preferred agents. Until the commercial availability of aztreonam-avibactam, aztreonam combined with ceftazidime-avibactam may be an optimal treatment option for S. maltophilia, including isolates resistant to levofloxacin and/or sulfamethoxazole-trimethoprim. Future studies with this combination against S. maltophilia are warranted.
********************************************************************************************************************
********************************************************************************************************************
********************************************************************************************************************
Spring Meeting Poster Abstracts
Original Poster Abstracts
ORIGINAL POSTER ABSTRACTS
On Site #1 – Original Research
Title: Operationalizing Dispensing Free Naloxone out of a Level 1 Trauma Center Emergency Department (ED)
Submitting Author: Rolla Sweis, PharmD, MA, BCPS
Authors: Diana Bottari, DO, Medical Director Pediatric Pain Management, Advocate Children's Hospital; Chris Boyle, PharmD, Clinical Pharmacist, Advocate Christ Medical Center; Meghan Soso, PharmD, BCPS, Clinical Manager, Advocate Christ Medical Center and Advocate Children's Hospital; Judith Brown-Scott, RPh, Operations Manager, Advocate Christ Medical Center and Advocate Children's Hospital; Mary Hormese, PharmD, BCPS, ED Pharmacist, Advocate Christ Medical Center and Advocate Children's Hospital; Marc McDowell, PharmD, BCPS, ED Pharmacist, Advocate Christ Medical Center and Advocate Children's Hospital; Rolla Sweis, PharmD, MA, BCPS, VP Operations, Advocate Christ Medical Center; Frank Belmonte, DO, MPH, VP Medical Management, Advocate Children's Hospital.
Organization: Advocate Christ Medical Center
Abstract
Purpose: Advocate Christ Medical Center in Oak Lawn, IL, is a 788-bed teaching institution. Advocate Christ is a leader in health care and is a major referral hospital in the Midwest. The hospital has one of the busiest Level I trauma centers in Illinois with more than 105,000 ED visits annually. As part of Advocate Health Care, Advocate Children’s is one of the largest pediatric services providers in the nation. Advocate Children’s Hospital in Oak Lawn has 35,000 ED visits annually.
The opioid crisis is a public health epidemic without boundaries. In 2017, in Illinois, 2,199 people died of opioid-related overdoses—nearly 2x the number of Chicago homicides and over 2x the number of car related deaths in the state reported that year. Since 2010, Advocate has served more than 30,000 patients for overdose or addiction treatment.
EDs are a major touch point in the opioid battle. After a non-fatal overdose, a patient is more likely to experience a fatal overdose. For every 1 fatal overdose there are an estimated 30 non-fatal overdoses. With inadequate treatment resources to treat the surge of patients, we are left to leave many patients on their own after an overdose. We wanted to assess the feasibility of dispensing free naloxone from one of our busiest EDs.
Methods: In partnership with a community program, Advocate Christ Medical Center and Children’s Hospital were able to obtain naloxone free of charge to both the hospital and patients through grants. Each kit provides 3 doses of 0.4mg naloxone, 3 intramuscular syringes and an outreach pamphlet. Lot numbers and expiration dates were documented in event of a recall. The risk department was consulted and approved all processes.
When a patient was identified (presented with overdose or opioid related event), or patient requested a naloxone kit, a “dummy” order was placed in the electronic medical record. A nurse, physician or ED pharmacist could place the order. The order triggered an outpatient label. The naloxone kit was labelled with all qualifiers required per Illinois dispensing laws. The labelled kit was then sent to the ED. The patient and/or family watched a brief video outlining how to identify an overdose, actions to take when an overdose has occurred, instructions to call 911, and how to administer naloxone. Dispensing of naloxone was reported to the Illinois prescription monitoring program.
Patients who refused to watch the instructional video did not receive the kit.
Results: In a 3-month period 40 kits were dispensed in the ED. One concern was increased volume of patients only wanting naloxone. However, this was not the case. We designed a handout in such an event which listed local pharmacies that participate in dispensing of naloxone without a prescription, including a pharmacy located within our hospital. An additional concern was the dispensing of the naloxone kit would delay discharge, but since all providers were educated on how to identify patients to offer the kit to, this did not occur.
Conclusion: As the opioid epidemic continues, we need to look at innovative ways to combat the crisis. Patients who die from an overdose are unable to go to rehabilitation. Most overdoses do not occur alone. Dispensing of the kit was not only to possibly save the patient’s life, but also potentially someone else’s. Dispensing of free naloxone from a level 1 busy tertiary hospital is feasible without adding to the wait times.
In conclusion, hospitals partnering with community outreach programs to combat the opioid crisis is one step in taming the crisis. Many community outreach programs have resources that hospitals do not. Many hospitals have touch points that community outreach programs do not. Combining our advantages has led to 120 doses of naloxone in our communities. The crisis is not one systems responsibility but all our responsibility.
********************************************************************************************************************
On Site #2 – Original Research
Title: Implementation of an enhanced recovery after surgery (ERAS) protocol in bariatric surgical patients to decrease post-operative nausea and vomiting: a retrospective chart review
Submitting Author: Kayla DuBois, PharmD, MBA
Authors: Kayla DuBois, PharmD/MBA, PGY1 Pharmacy Practice Resident, Memorial Medical Center; Jennifer Ratliff, RPh, BCPS, Memorial Medical Center; Orlando Icaza, Jr., MD, FACS, Memorial Medical Center; Tiffany Turner, APRN, MSN, CBN, Memorial Medical Center; Don Ferrill, PharmD, BCPS, Memorial Medical Center.
Organization: Memorial Medical Center
Abstract
Purpose: Nausea and vomiting in the post-operative period is a significant concern for both patients and providers that leads to greater patient dissatisfaction and contributes to increased hospital length of stay and morbidity. Decreasing post-operative nausea, vomiting, and opioid use is a priority among health care providers. This study aims to enhance the small pool of currently available published literature and provide evidence of a novel enhanced recovery after surgery (ERAS) program in bariatric surgical patients. This ERAS program employs a pre-operative regimen of aprepitant, transdermal scopolamine, acetaminophen, pregabalin, and celecoxib to decrease post-operative antiemetic and opioid requirements.
Methods: This study has gained approval from the local Institutional Review Board. The retrospective chart review will identify patients who underwent bariatric surgeries, including roux-en-y gastric bypass and sleeve gastrectomy, and compare outcomes pre- and post-implementation of the ERAS program. Exclusion criteria include any patient in the post-implementation group in which the ERAS protocol was deviated from, patients who underwent revision of a previous bariatric procedure, and patients on chronic opioid therapy prior to the procedure. The following data will be collected for analysis: age, gender, length of hospital stay, procedure start and end time, Bariatric Risk Score, American Society of Anesthesiologists Physical Status score, allergies, intra-operative drug administration, route of anesthesia, chronic opioid use, serum creatinine, pain scores, post-operative antiemetic use, and post-operative opioid use. The primary outcome will be post-operative anti-emetic use, measured as the number of individual doses of antiemetic agents and the number of individual antiemetic agents administered in the post-operative period (cumulatively at 24 hours, 48 hours, and total period). Secondary outcomes will include post-operative opioid use measured as total morphine milligram equivalents administered in the post-operative period (cumulatively at 24 hours, 48 hours, and total period), hospital length of stay, and cost analysis.
Results: Presented at meeting
Conclusions: Presented at meeting
********************************************************************************************************************
On Site #3 – Original Research
Title: Impact of pharmacy process improvements within the emergency department on time to antibiotic administration and outcomes in patients with sepsis
Submitting Author: Katelyn Fryman, PharmD
Authors: Katelyn Fryman, PharmD, Memorial Medical Center; Megan Metzke, PharmD, BCPS, BCCCP, Memorial Medical Center; Michael Guithues, PharmD, BCPS, Memorial Medical Center; Julie Downen, PharmD, BCPS, CLSSBB, Memorial Medical Center; Don Ferrill, PharmD, BCPS, Memorial Medical Center; Trupesh Chanpura, MD, Memorial Medical Center.
Organization: Memorial Medical Center
Abstract
Purpose: The surviving sepsis campaign recently released new guidelines which recommend initiating intravenous antibiotics as soon as possible, not to exceed one hour after recognition of sepsis. The objective of this study is to implement new pharmacy processes to decrease time to antibiotic administration, as well as evaluate the effect these processes had on patient outcomes.
Methods: This study was approved by the local Institutional Review Board. Patients will be identified by initiation of the sepsis order set in the electronic medical record system. Exclusion criteria will include patients that transferred from an outlying hospital, patients who did not receive antibiotics within the first six hours, patients missing time of antibiotic administration, patients receiving antibiotics prior to sepsis presentation, and patients without suspected or diagnosed sepsis. Both a retrospective and prospective review of these patient charts will be completed to determine if there are any significant changes in patient outcomes after implementation of new processes. New processes include an alert to pharmacists when a patient has suspected sepsis, stocking commonly prescribed antibiotics for sepsis in the automated dispensing machine for removal, and education to nursing staff in the emergency department on the importance of timely antibiotic administration. The following data will be collected: patient demographics, time to antibiotic initiation, mortality, hospital length-of-stay, intensive care unit length-of-stay, and duration of vasopressor use. All data will be stored and collected on secure health system computers. Data collection documents will be encrypted, and password protected to prevent exposure of data to unintended persons.
Results: Presented at meeting
Conclusion: Presented at meeting
********************************************************************************************************************
On Site #38 – Original Research
Title: Value of pharmacy technician clinical support role in primary care
Submitting Author: Emily Hoadley, PharmD
Authors: Emily M. Hoadley, PharmD, PGY-2 Ambulatory Care Pharmacy Resident, Northwestern Memorial Hospital; Bridget M. Dolan, PharmD, Clinical Pharmacist, Northwestern Medicine Specialty Pharmacy; Amy E. Wainright, PharmD, BCACP, BCGP, Clinical Pharmacist, Northwestern Medicine Specialty Pharmacy; Joshua J. Wentland, CPht, Pharmacy Technician Specialist, Northwestern Medicine Specialty Pharmacy.
Organization: Northwestern Memorial Hospital
Abstract
Purpose: A pharmacy technician practicing at the top of their license is a valuable asset that supports medication optimization and access, including but not limited to, identification of patients who would benefit from additional medication or disease state education and financial assistance. A clinical support pharmacy technician has the potential to increase the reach and impact of clinical pharmacists in a cost-effective manner. At Northwestern Medicine, a pharmacy technician was added to the Care Coordination team, composed of nurses, social workers, and a pharmacist, to support outpatient primary care clinics in May 2018. Evaluating the success of this position may set a valuable precedent for a non-dispensing pharmacy technician role in team-based care. The purpose of this project is to describe the role and assess the interventions of a pharmacy technician added to the Care Coordination team.
Methods: The role, workflow, and responsibilities of the pharmacy technician will be obtained via interview and described. Care Coordination team members will be surveyed to assess their perception of the role of the pharmacy technician and the value of this role in Care Coordination. A retrospective review of all interventions and patient interactions recorded by the Care Coordination pharmacy technician from May 2018 to December 2018 will be completed. Descriptive statistics will be used to determine the type, quantity, and outcome of the interventions. Additionally, cost savings to the patient, when applicable, will be reported.
Results: The pharmacist and pharmacy technician identified the workflow and responsibilities for the role. Referrals to the pharmacy technician came from the Care Coordination team, primary care providers outside of the Care Coordination team, and quality metric reporting. Responsibilities included: performing medication formulary investigation, obtaining insurance coverage and financial assistance by completing forms for prior authorizations and patient assistance programs, working with the pharmacist to provide recommendations for alternate covered therapies and relaying the information back to the prescribers and staff, and triaging patients to the pharmacist for clinical intervention or education. A majority of the interventions were related to medications for diabetes and interventions took the pharmacy technician a median of 30 minutes to complete. From May to December 2018, the technician reported a total of 608 interventions completed or ongoing. These interventions resulted in cost savings to our patients of at least $50,000 per month.
Conclusions: A clinical support pharmacy technician was successfully added to the Care Coordination team at Northwestern Medicine in May 2018. The pharmacy technician role has provided significant financial benefit to our patients. Clearly defining the role, responsibilities, and scope of practice of the pharmacy technician in a primary care setting will allow for pharmacy technicians and pharmacists to practice at the top of their licenses. Future addition of clinical support pharmacy technicians to multidisciplinary teams in primary care is a cost effective way to extend the reach of clinical pharmacy services.
********************************************************************************************************************
On Site #39 – Original Research - Winner Best Original Poster
Title: Cost Savings Associated with a Therapeutic Conversion of Insulin Aspart to Insulin Lispro
Submitting Author: Sarah Kessler, PharmD
Authors: Sarah G. Kessler, PharmD, PGY-2 Internal Medicine Pharmacy Resident, University of Chicago Medicine; Jennifer Austin Szwak, PharmD, BCPS, Clinical Pharmacist Specialist, Internal Medicine PGY2 Internal Medicine Residency Program Director, University of Chicago Medicine; Hailey P. Soni, PharmD, BCPS, Clinical Pharmacist Specialist, Internal Medicine, PGY1 Pharmacy Residency Program Coordinator, University of Chicago Medicine; Randall Knoebel, PharmD, BCOP, PGY1 Pharmacy Residency Program Director, Senior Manager, Pharmacy Health Analytics & Drug Policy Director, Pain Stewardship Pharmacy, University of Chicago Medicine.
Organization: University of Chicago Medicine
Abstract
Purpose: As drug prices continue to increase affecting all areas of patient care in which medications are administered or dispensed, identifying avenues for reducing costs has become critical. One avenue hospitals can control their drug expense is through optimization of procurement practices, including examining purchases on wholesale acquisition cost (WAC) to identify unnecessary spending. Insulin aspart, our formulary rapid acting insulin, had a high percentage of its spend falling to the WAC account. Insulin aspart is available only in 10mL vials and 3mL Flexpens, resulting in a significant amount of unused drug due to short dating of product and stocking procedures on the floor. The purpose of this study was to determine whether a therapeutic interchange from insulin aspart to insulin lispro would result in decreased WAC expenditure and overall cost savings.
Methods: In an effort to minimize waste and reduce WAC expenditure, the Pharmacy Department shifted its formulary rapid acting insulin product from insulin aspart to insulin lispro to capitalize on a smaller vial size of 3mL. In order to do this, current supply of insulin aspart was short dated, and insulin lispro was pre-loaded into the automated dispensing cabinets. Insulin lispro was inaccessible until the day of implementation. On the day of implementation, all new orders for rapid acting insulin defaulted to insulin lispro. Concurrently, pharmacists converted previously active insulin aspart orders to insulin lispro. The primary outcome of this study was to determine whether this conversion would result in a reduction in insulin expenditure.
Results: The average monthly cost of rapid acting insulin prior to changing from insulin aspart to insulin lispro was roughly $13,000 per month. After changing to insulin lispro, the average monthly cost of rapid acting insulin decreased to roughly $7,000. This resulted in a cost savings of about $5,000 per month. Additionally, a large decrease in the percentage of expenditure falling to WAC was noted. Further reduction in average cost per month is expected as costs become more stable over time.
Conclusions: By converting the formulary insulin from 10mL insulin aspart vials to 3mL insulin lispro vials, the University of Chicago Medicine was able to capitalize on both reduced cost and reduced WAC by reducing waste. The pharmacy department hopes to continue pursuing this form of procurement practice with additional insulin formulations.
********************************************************************************************************************
********************************************************************************************************************
********************************************************************************************************************
Spring Meeting Poster Abstracts
Encore Poster Presentations
ENCORE POSTER ABSTRACTS
On Site #4 – Encore
Title: The Financial Impact of Pharmacist Interventions in an Ambulatory Care Oncology Clinic
Submitting Author: Taylor Conklin, PharmD
Authors: Taylor Conklin, PharmD, Northwestern Memorial Hospital; Alison Svoboda, PharmD, Northwestern Memorial Hospital; Farah Barada, PharmD, Northwestern Memorial Hospital; Chelsea Gustafson, PharmD, Northwestern Memorial Hospital; Caitlin Bowman, PharmD, Northwestern Memorial Hospital.
Organization: Northwestern Memorial Hospital
Abstract
Purpose: The role of oncology clinical pharmacists has continued to evolve in the past decade, as there has been a transition from inpatient management to outpatient management through ambulatory care clinics. A number of previous studies have determined a significant benefit of incorporating a clinical pharmacist into these ambulatory oncology clinics; however, very few studies justify pharmacists’ impact through revenue opportunities and cost avoidance. Randolph et al.’s pilot study on cost avoidance in an ambulatory oncology clinic uncovered the need for more full-time clinical pharmacists in the outpatient setting. Their interventions could be extrapolated to an estimated annual cost avoidance of $282,741 per pharmacist per year. Northwestern Memorial Hospital currently has implemented one full time equivalent devoted to its outpatient oncology clinic. This pharmacist has been embedded within the care teams specifically working with GI and melanoma patients. Additionally, physicians have started to bill at a higher tier when incorporating a clinical pharmacist in their model. This presents opportunities for both cost avoidance and a potential new source of revenue within the clinic. The purpose of this study was to determine the impact of clinical pharmacists in the ambulatory oncology clinic through cost avoidance and opportunity for revenue capture.
Methods: This prospective, observational study took place during a four week period. The clinical pharmacist documented four categories of interventions and corresponding time spent per intervention in their daily workflow: chemotherapy regimen review, medication teaching, supportive care, and medication reconciliation. The cost avoidance values assigned to these interventions were compiled from previous literature and updated for current inflation for 2018. An analysis of tier billing was collected during this study period to identify gaps in revenue opportunities within the GI and melanoma practice.
Results: A total of 205 interventions were documented during this four-week period. The most frequent intervention made by clinical pharmacists was medication reconciliation (n = 74), followed by supportive care (n = 54). Cost avoidance per intervention was calculated by multiplying the time spent for intervention by cost-avoidance values outlined in previous literature. The cost avoidance calculated during the study period was $21,403. This number was extrapolated to a 40 hour work week with 52 weeks per year, and was calculated to be $278,309. The analysis of tier billing determined the estimated annual revenue captures missed by not billing a pharmacist during patient appointments to be $52,427. This opportunity cost for revenue capture was added to the annual net benefit. The estimated cost of employing a clinical pharmacist in the oncology clinic was $116,310. Subtracting this cost from the annual cost avoidance yielded a net benefit of $214,356 per pharmacist.
Conclusion: This study concluded that full-time clinical pharmacists are financially beneficial to ambulatory oncology clinics. Given the financial impact, adding more full-time equivalent pharmacists may provide more revenue benefits and potential income growth.
********************************************************************************************************************
On Site #5 – Encore
Title: Student Pharmacist Driven Medication History Training Pilot Program
Submitting Author: Haley Fox, PharmD Candidate
Authors: Haley M. Fox, PharmD Candidate; Amanda Kojda, PharmD Candidate; Lauren Strombolic, PharmD Candidate; Denise Lamm, PharmD Candidate; Omar Jaber, PharmD Candidate; Andrew Mclnerney, PharmD Candidate; Sophia Gilardone, PharmD Candidate; Rolla Sweis, Advocate Christ Medical Center, PharmD, MA, BCPS; Marc McDowell, Emergency Medicine Pharmacist, Advocate Christ Medical Center, PharmD, BCPS.
Organization: Advocate Christ Medical Center
Abstract
Purpose: To pilot a medication history program in a high volume emergency department using experienced pharmacy students to train novice students. The Emergency Department (ED) at Advocate Christ Medical Center (ACMC) is a 120 bed level 1 trauma center. Obtaining medication histories is an essential process for appropriate pharmacotherapy management of patients being admitted from the ED. Typically, this function is performed by nursing staff; however, high rates of errors have been observed, which may lead to worse patient outcomes. Evidence suggests student pharmacists can effectively obtain medication histories and avoid potential adverse drug events. We sought to explore utilizing experienced student pharmacists to train novice student pharmacists in obtaining accurate medication histories in a high capacity hospital such as ACMC.
Methods:- Seven student pharmacists were recruited for this project including three experienced students and four novice students. Experienced students participating in the program required at least 120 hours of previous ED medication history experience. The novice students had no prior experience in obtaining medication histories. All student pharmacists underwent a six-hour training process with an ED pharmacist. Students were educated on the basics of the electronic medical record (EMR) and underwent a basic review of how to perform medication histories. Subsequently, each student performed a medication history and were observed by an ED pharmacist. For two weeks, the experienced students led and supervised the novice students obtaining medication histories. The final week of the pilot program, experienced and novice students practiced independently. The ED pharmacist reviewed all medication histories for accuracy and completeness. Patients were identified as requiring a medication history if an admission request was placed in the EMR. If all patients being admitted had their medication histories performed, students were instructed to proactively acquire medication histories based on the Emergency Severity Index Tool, a five-level triage urgency scale. This pilot program evaluated all patients in the ACMC ED from December 26th, 2017 to January 13th, 2018.
Results: Over a span of three weeks, the seven pharmacy students participating in the medication reconciliation pilot program completed 466 medication histories. Twice weekly, each novice student was paired with an experienced student and participated in eight hour shifts. These teams obtained medication histories under the direction of the experienced students for the first two weeks of the program. During the training weeks, an average of 18.08/24 medication histories were completed during each eight hour shift. After completing two weeks of training, novice and experienced students obtained medication histories independently for one week. On average, the experienced students completed 22.17 medication histories per eight hour shift, while the novice students completed an average of 13.25 medication histories per 8 hour shift. Once completed, a 15% sample size of all medication history was reviewed by an ED pharmacist for accuracy and completeness. Correct drug, frequency, route, dose, dosage form, and patient compliance were evaluated. Experienced students had an overall accuracy rate of 97.9% compared to novice students 96.8% when practicing independently. Across all students, modest improvements were observed in all categories.
Conclusions: Through this program, novice pharmacy students were able learn how to complete accurate medication histories in a timely manner in the ED. The results did show that novice students were able to complete fewer medication histories per shift; however medication histories completed by the novice students were as accurate as experienced students and speed would likely be gained with experience. Overall, a student precepted medication history pilot service was an effective program in the training of novice students to accurately complete medication histories.
********************************************************************************************************************
On Site #6 – Encore
Title: Assessing current gaps in practice and improving hospital adherence to ISMP's Best Practice recommendations surrounding oral methotrexate use in non-cancer indications
Submitting Author: William Justin Moore, PharmD
Authors: William Justin Moore, PharmD, PGY1 Pharmacy Resident, Northwestern Memorial Hospital; Justin Daniel Fisher, PharmD, PGY2 Pharmacy Resident, Northwestern Memorial Hospital; Daniel J. Wojenski, PharmD, BCOCP, Hematology/Oncology Practice Coordinator, Northwestern Memorial Hospital.
Organization: Northwestern Memorial Hospital
Abstract
Purpose: Medication errors associated with methotrexate have been reported to include nearly every identifiable facet of patient care, including prescribing, dispensing, patient education, and medication reconciliation. With an array of therapeutic indications including those of oncologic and non-oncologic use, the largest obstacle in addressing medication errors related to methotrexate surrounds the frequency of dosing. Methotrexate was added to the Institute for Safe Medication Practices’ (ISMP) List of High-Alert Medications in 2003 in an effort to curtail avoidable oversight. Reports of adverse or fatal errors have been featured over 60 times in ISMP’s Medication Safety Alert! Newsletters since 1996. The vast majority of these reports include oral methotrexate being prescribed for the treatment of non-oncologic disease states with many errors resulting in hospitalizations and patient deaths. The inconsistent practice surrounding methotrexate in non-oncologic indications is a multi-faceted issue facing clinicians and patients across the continuum of care. Despite an abundance of case reports highlighting preventable and sometimes fatal healthcare errors, awareness and adherence to ISMP Best Practice recommendations regarding methotrexate is disproportionately low. Among 501 US hospitals who provided self-assessments of compliance to ISMP recommendations only 46% reported full implementation of the seven recommended best practices. Pharmacists play a vital role in the stewardship of medication safety to ensure patients receive appropriate therapy while minimizing potential harm. This project aims to evaluate our institution’s adherence to ISMP’s Best Practice recommendations surrounding methotrexate for non-oncologic indications in an effort to prevent adverse medication errors and improve patient care.
Methods: Admitted patients with active orders for oral methotrexate were analyzed including those orders placed from June 1, 2018 through September 15, 2018. Data points collected included indication, dose, frequency, route of administration instructions, and prescriber specialty. In addition, all outpatient orders for oral methotrexate prescribed during the same time period were also analyzed. Data collected included number of tablets prescribed, duration of therapy as days supplied, and administration instructions. Each inpatient and outpatient order was considered to be “compliant” if it met all criteria listed in the ISMP Best Practice Recommendations or “noncompliant” if it did not. Orders were evaluated based on each of the seven ISMP recommendations individually and comprehensively.
Results: At our institution oral methotrexate prescribing does not consistently adhere to ISMP Best Practice Standards. Rheumatoid arthritis accounted for the majority of uses, with 64% and 57% of inpatient and outpatient orders, respectively. Among the seven Best Practice recommendations our institution was 100% adherent to only two of the seven standards. We identified the following gaps: inadequate discharge education and counseling including patient comprehension strategies via verbal and written education; lack of uniform prospective auditing of dosing regimens performed by healthcare professional prior to discharge or administration; inability to place order with day-specific administration instructions; a lack of limitations surrounding maximum days of prescribed therapy; inconsistent ordering practices regarding verification of the ordering provider as an oncologist.
Conclusions: As a result of this investigation, gaps were identified and addressed in order to improve medication safety by better adhering to ISMP recommendations through IT improvements and optimization of patient discharge counseling. Various quality initiatives will be implemented to address these gaps in practice to ensure safe and quality healthcare is delivered to those patients receiving methotrexate for non-oncologic indications. This project may serve as a reference for other institutions to identify gaps in practices relating to quality and safety surrounding methotrexate in this setting of use in order to limit preventable medication errors and provider oversight. There is a dire need for continued implementation of quality initiatives across health-systems to reduce the occurrence of medication errors with methotrexate.
********************************************************************************************************************
On Site #7 – Encore
Title: Impact of Team-Based Learning on Pharmacy Students’ Knowledge and Confidence in Searching Primary Literature
Submitting Author: Elana Nelson, PharmD
Authors: Elana M. Nelson, PharmD, Instructor/Laboratory Instruction Coordinator, Pharmacy Practice, Department of Biopharmaceutical Sciences, University of Illinois at Chicago College of Pharmacy; Samantha Spencer, PharmD, Clinical Assistant Professor, Pharmacy Practice, Drug Information Group, University of Illinois at Chicago College of Pharmacy; Courtney D. Krueger, PharmD, Clinical Assistant Professor, Pharmacy Practice, Drug Information Group, University of Illinois at Chicago College of Pharmacy.
Organization: University of Illinois at Chicago College of Pharmacy
Abstract
Purpose: To evaluate the impact of team-based learning (TBL) on knowledge and confidence in primary literature searching skills for pharmacy students.
Methods: TBL was integrated into a drug information course to teach primary literature searching skills. Previously, a traditional lecture format was used. To assess the impact of TBL, students instructed in both formats were invited to participate in an online survey that assessed perceived confidence and primary literature searching knowledge one year after completion of the course. Survey results were compared between cohorts.
Results: 42 and 63 pharmacy students in the TBL and traditional lecture cohort, respectively, completed the survey. For knowledge assessment questions, no statistically significant difference in scores existed between cohorts. 100% of students in the TBL cohort reported the course improved their ability to search primary literature versus 94% in the traditional lecture cohort (p=0.034). For the primary literature recitation, the TBL format differed substantially from the delivery in the traditional cohort and 90% of students in the TBL cohort agreed or strongly agreed that the recitation improved their ability to search primary literature compared with 79% in the traditional lecture cohort (p=0.016).
Conclusions: Our findings are consistent with previous research showing that incorporating TBL into drug information courses can have a positive effect on confidence. Further research is needed to assess the long-term impact and sustainability of TBL on knowledge retention and clinical application of primary literature searching skills during student clerkships and future professional practice.
********************************************************************************************************************
On Site #40 – Encore – Winner Best Encore Poster
Title: Comparing the efficacy of serum vancomycin concentrations of pharmacist-driven versus infectious disease physician-driven dosing of vancomycin
Submitting Author: John Talili, PharmD Candidate
Authors: John A. Talili; PharmD Candidate, Southern Illinois University Edwardsville School of Pharmacy; Elizabeth A. Cady, PharmD, BCPS, Southern Illinois University Edwardsville School of Pharmacy; Maithili Deshpande, PhD, Southern Illinois University Edwardsville School of Pharmacy; Natalie R. Tucker, PharmD, BCPS, HSHS St. John's Hospital.
Organization: Southern Illinois University Edwardsville School of Pharmacy
Abstract
Purpose: Vancomycin, an antibiotic, is primarily used in the treatment of methicillin-resistant Staphylococcus aureus infections. The dosing/monitoring of vancomycin has been widely adopted by clinical pharmacists, via protocols and collaborative practice agreements. However, physicians may take the responsibility of dosing/monitoring. It is unknown whether pharmacists or specifically, infectious disease-trained physicians, can more safely and effectively dose vancomycin. The purpose of this retrospective study was to compare 1. the percentage of time vancomycin troughs were within range and 2. the average number of vancomycin levels ordered per day of vancomycin therapy between ID physicians and pharmacists within an institution.
Methods: This study was a retrospective chart review and was approved by the institutional review board. The study included patients who were 18 years of age and/or older. The dates of the review were arbitrarily chosen from March 19, 2018 to August 5, 2018. Patients were included if they had been treated with vancomycin. At this 431-bed hospital, protocol states that all vancomycin is to be dosed and monitored by a pharmacist, unless the patient is under the care of the ID consult service. In this case, the ID physician will dose and monitor the vancomycin. Patients were grouped into two categories with their vancomycin being dosed by either 1. a pharmacist or 2. an infectious disease physician. Patients were excluded if they were below 18 years of age. The primary outcome was the percentage of vancomycin serum drug levels (defined as troughs and random levels) within goal range (in mcg/mL), as dosed by either pharmacist or infectious diseases physician. Goal ranges were determined based on the patient’s infectious disease diagnosis. Secondary endpoints were the average number of vancomycin levels per day drawn and average number of days patients were on vancomycin therapy.
Results: The sample consisted of 30 patients (15 in each group), of which 16 were male and 14 were female. The mean age was 63 years old with 6 patients (three from each group) on hemodialysis during the study period. A total of 151 vancomycin levels were drawn in this patient population. Pharmacists had a total of 65 vancomycin levels drawn with 29 levels within range (45%) while infectious disease physicians had a total of 86 levels drawn with 42 levels within range (49%). Between the pharmacist and infectious disease physician dosing groups, there was no statistically significant difference in vancomycin levels within range (p = 0.61). In terms of average number of vancomycin levels drawn, pharmacists and infectious diseases physicians were similar with 0.62 and 0.51 respectively with no statistical difference (p = 0.65). Patients were on vancomycin for an average of 7 days (the longest of which was on vancomycin for 17 days) with pharmacist dosing and 11.2 days (the longest of which was on vancomycin for 48 days) with infectious diseases dosing with no statistical difference between the two groups (p = 0.22).
Conclusions: In this retrospective study, pharmacists and infectious diseases physicians had similar efficacy in terms of dosing vancomycin within goal range (troughs and random levels) and in terms of number of vancomycin levels drawn per day of therapy. Limitations include a small sample size and the fact that an ID pharmacist often recommends vancomycin doses/monitoring on the ID consult service. A larger study with more patients included must be conducted to optimally assess outcomes and to successfully determine which team is more effective at dosing and monitoring vancomycin, whether it be pharmacists or infectious disease physicians.
********************************************************************************************************************
********************************************************************************************************************
********************************************************************************************************************
Spring Meeting Poster Abstracts
Student Research Poster Abstracts
STUDENT RESEARCH POSTER ABSTRACTS
On Site #8 – Student Research
Title: Concurrent use of benzodiazepines and opioids: a drug utilization review-based investigation in a community hospital
Submitting Author: Ashley Riley, PharmD Candidate
Authors: Ashley M. Riley, PharmD candidate, Southern Illinois University Edwardsville; Kristi D. Stice, PharmD, BCPS, Executive Director of Pharmacy and Quality Systems, Decatur Memorial Hospital.
Organization: Southern Illinois University Edwardsville School of Pharmacy
Abstract
Purpose: Concurrent use of benzodiazepines and opioids places patients at increased risk of oversedation. Pharmacists are in a unique position to be able to provide prescribers with drug expertise, contributing to safe and optimal patient care. Through utilization of intervention tools and electronic health system reports, pharmacists can identify patients who are at risk for oversedation events related to concurrent benzodiazepine and opioid therapy. The goal of this retrospective drug utilization review-based quality improvement project is to improve patient outcomes and maximize patient safety related to opioids and benzodiazepines in the hospital setting.
Methods: Drug utilization reports specific to the medical and surgical nursing floors of a single hospital site were obtained. Data collected included the number of patients prescribed opioids and benzodiazepines concurrently, the number of patients prescribed opioids, the number of naloxone reversals in patients prescribed opioids, and the number of pharmacist interventions related to concurrent use. Prescriber and pharmacist education was presented and a report was constructed within the electronic health record system to help pharmacists identify patients on concurrent opioid and benzodiazepine therapy. Pharmacists were educated on how to generate the report and were encouraged to track interventions made addressing concomitant medication orders. Data was collected before and after implementation of the electronic health record report and prescriber education.
Results: A total of 70 patients were on concurrent opioid and benzodiazepine therapy in October 2017. A total of 60 patients were on concurrent opioid and benzodiazepine therapy in November 2017. Concurrent drug utilization decreased in 2018, with records of 51 patients in October and 36 patients in November. The surgical nursing floor had higher incidence of concurrent use compared to the medical nursing floor. A total of 20 pharmacist interventions were recorded in 2018 specific to the surgical nursing and medical nursing hospital floors. A total of 705 patients were prescribed opioids during their hospital stay in 2017 compared to 678 patients in 2018. Naloxone administrations decreased from 6 administrations in 2017 to zero administrations in 2018. Overall, there needs to be more studies conducted to determine the relationship between pharmacist methods of communication for intervention and changes in prescribing patterns.
Conclusions: There was a clinically significant reduction in the number of patients on concurrent opioid and benzodiazepine therapy, which was mirrored by a reduction in naloxone reversals as well. These findings suggest that opioid and benzodiazepine stewardship may play a role in reducing adverse events and increasing patient safety overall.
********************************************************************************************************************
On Site #9 – Student Research
Title: A Retrospective, Single Center Study Evaluating COPD Management and Hospital Readmissions
Submitting Author: Lauren Ballweg, PharmD Candidate
Authors: Lauren Ballweg, PharmD Candidate; Carrie N. Vogler, PharmD, BCPS, SIU School of Medicine.
Organization: Southern Illinois University Edwardsville
Abstract
Purpose: Chronic Obstructive Pulmonary Disease (COPD) contributes to 5% of global deaths and billions in health care spending. The increased attention of health care spending and the push towards value-based services has lead Centers for Medicare and Medicaid Services (CMS) to take a closer look at patients with COPD, and the rates of hospital admissions and readmissions. This shift in focus comes with the intent of improving quality of life, decreasing exacerbations and disease progression, and provides a role for pharmacists to ensure proper medication prescribing and utilization. The GOLD Guidelines provide recommendations for COPD medications throughout different stages of disease progression; initial treatment, step up therapy and exacerbations. The primary objective of this study is to assess the hospital discharge medications for COPD after a hospitalization due to an exacerbation. This study will evaluate the COPD medications used and the recurrence of hospital readmission rates among this population.
Methods: The study was conducted via retrospective chart review of patients admitted to SIU Internal Medicine Hospitalist services at Memorial Medical Center between December 1, 2017 and July 1, 2018. Eligible patients were identified by ICD 10 codes indicative of COPD or acute COPD exacerbation. Once participants were identified the medications used for the management of COPD were assess and compared to the recommendations of the GOLD 2018 Guidelines. Primary outcomes included 30 day all cause readmission rates for patients with documented COPD; COPD medications prescribed at discharge and the appropriateness of dosing compared to GOLD guidelines; and hospitalization due to COPD exacerbation.
Results: Presented at meeting
Conclusions: Presented at meeting
********************************************************************************************************************
On Site #10 – Student Research
Title: Does The Infusion Time of IVIG Matter in Kawasaki Disease?
Submitting Author: Joseph Griffin, PharmD Candidate
Authors: Joseph Patrick Griffin, PharmD Candidate, Chicago State University; Angela Powell, PharmD Candidate, Chicago State University; Shannon Rotolo, PharmD, Clinical Pharmacist, University of Chicago Medicine; Palak Bhagat, PharmD, Clinical Pharmacist, University of Chicago Medicine; Allison Bartlett, MD, Associate Professor Pediatrics, Section of Infectious Diseases, University of Chicago Medicine.
Organization: Chicago State University / University of Chicago Medicine
Abstract
Purpose: Evaluate if there is a difference in the incidence of coronary aneurysms in those who received an Intravenous Immune Globulin (IVIG) 2g/kg dose over 10-12 hours as compared to those who received a dose over less than 10 hours.
Methods: Patients diagnosed and treated for Kawasaki Disease with IVIG at University of Chicago Medicine will be determined by drug utilization reports. The reports will include patients who received IVIG between September 1, 2008 and August 31, 2018. Data will be collected through chart review and patients will be divided into two groups based on duration of infusion (less than 10 hours or 10-12 hours). The primary endpoint will be the incidence of coronary aneurysms. The secondary endpoint will be the resolution time of acute phase reactants and defervescence. The safety endpoints will measure the risk of thrombosis and renal dysfunction.
Results: Presented at meeting
Conclusions: Presented at meeting
********************************************************************************************************************
On Site #11 – Student Research
Title: A comparison of renal outcomes among targeted immunotherapies
Not presented – Results not available at time of meeting.
On Site #12 – Student Research
Title: Analyzing the Role of Pharmacists in Overcoming Inadequate Documentation of Angiotensin Converting Enzyme Inhibitor Induced Drug Reactions in the Electronic Medical Record (Updated title)
Submitting Author: Myesha Tabriz, PharmD Candidate
Authors: Myesha Tabriz, PharmD Candidate, University of Illinois at Chicago (UIC) College of Pharmacy; Reham Awad, PharmD Candidate, University of Illinois at Chicago (UIC) College of Pharmacy; Alvin Godina, PharmD, BCPS, Roosevelt University/Advocate Christ Medical Center; Marc McDowell, PharmD, BCPS, Advocate Christ Medical Center.
Organization: University of Illinois at Chicago College of Pharmacy / Advocate Christ Medical Center
Abstract
Purpose: Angiotensin Converting Enzyme Inhibitor (ACE-I) induced drug reactions such as a dry cough or angioedema are common drug related adverse reactions experienced by patients. Improper documentation of such drug related adverse events, or the lack thereof, may pose severe or life threatening risks to patients if left unnoticed by health care providers. Therefore, the objective was to identify the prevalence of undocumented or inadequately documented ACE-I induced reactions in the banner of the electronic medical record (EMR). Furthermore, the chart review will examine the accuracy of medication induced drug reaction documentation and will assess the role of pharmacists’ in improving proper documentation in the EMR banner.
Methods: The charts of all patients enrolled in the Advocate Christ Medical Center (ACMC) outpatient congestive heart failure (CHF) clinic were reviewed from December 18, 2018 to January 11, 2019. Initially, the patient banner in Cerner PowerChart was assessed for documentation of an ACE-I adverse reaction. Subsequently, the patient chart was reviewed utilizing the “chart search” function with key words such as “ACE-I” for instances of reported ACE-I adverse effects elsewhere in the EMR. The data that was collected included whether or not the patient experienced an ACE-I adverse reaction, whether or not it was documented correctly, and if it was documented by a pharmacist. Correct documentation was assessed based on proper listing of the drug or drug class that caused the adverse effect, followed by what the adverse effect was, and when the adverse effect occurred.
Results: There was a total of 804 patients that were enrolled in the ACMC outpatient CHF clinic that were analyzed in this study. Of the 804 patients, 117 patients had an unintended side effect to an ACE-I such as a dry cough and angioedema. Of the 117 patients, 38 cases had correct documentation of a noted adverse effect to an ACE-I in the banner of the EMR, 39.47% of which were documented by a pharmacist. Additionally, there were 43 cases of inadequate documentation of ACE-I induced adverse reactions with only 9.3% being documented by a pharmacist. Inadequate documentation comprised of an ACE-I reported in the banner as a warning without indicating the adverse reaction that the patient experienced or the onset. Moreover, there were 36 cases of ACE-I induced adverse reactions that were present in the patient’s chart; however, were not documented in the banner. The 36 cases that were not documented in the EMR banner comprise 30.77% of the total allergies that were found among the heart failure patients that were analyzed.
Conclusion: Overall, there is a higher incidence of ACE-I induced adverse reactions that were mis-documented or not documented compared to those that were correctly documented which allows us to conclude that improvements must be implemented in order to achieve proper documentation in the EMR. Unfortunately, there is a lack of available guidelines for proper documentation of allergies and significant adverse reactions within patients’ health record. Therefore, future implications from this study include the standardization of allergy documentation for all healthcare professionals to follow in order to overcome the discrepancy and errors observed in the current system due to the higher incidence of inadequate documentation completed by healthcare professionals outside the field of pharmacy.
Additionally, based on the results, it is evident that pharmacists do not currently play a major role in documentation of drug induced reactions in the EMR. Over the years, pharmacists have gained the role of completing medication reconciliation interviews; therefore, pharmacists’ role should expand to include documentation of patient allergies and drug induced reactions. The role expansion could help overcome issues that were observed in the EMR such as missing information of the drug-induced reaction, time that the reaction occurred, as well as incorrect information being documented under the allergy section of the EMR banner.
If we were to improve the study, a larger population would be analyzed to further solidify our conclusion. Additionally, the data analysis would include the quantification of re-prescribing of an ACE-I in the incidents of correct, incorrect, or mis-documentation as a measure of patient safety. Lastly, the study would further support the need for pharmacists to be responsible for conducting medication reconciliation interviews and documenting it in the EMR. To conclude, this study calls for a standardization of allergy and drug induced reaction documentation as well as the expanded role of the pharmacist in this setting.
********************************************************************************************************************
On Site #41 – Student Research
Title: Nalbuphine as a Primary Parenteral Opioid for Acute Pain in a Large Community Hospital: A Retrospective Review.
Submitting Author: Danielle Vahlkamp, PharmD Candidate
Authors: Danielle Vahlkamp, PharmD Candidate, Southern Illinois University Edwardsville School of Pharmacy; Amanda Daniels, PharmD, BCPS, HSHS St. Elizabeth's Hospital; Chris Herndon, PharmD, FASHP, Southern Illinois University Edwardsville School of Pharmacy.
Organization: Southern Illinois University Edwardsville School of Pharmacy
Abstract
Purpose: To determine if intravenous (IV) nalbuphine could be a reasonable, alternative parenteral analgesic option to treat acute pain in a hospital when compared to first-line pain medications such as intravenous morphine during drug shortages.
Methods: This retrospective chart review occurred at HSHS St. Elizabeth’s Hospital in O’Fallon, IL between January 1, 2018 and June 30, 2018. Patients admitted to HSHS St. Elizabeth’s Hospital who received IV morphine or IV nalbuphine for the treatment of acute pain were included in analysis. The primary outcome was the average pain score during an individual hospital stay. Secondary outcomes consisted of required change in parenteral opioid during hospital stay, opioid presence at admission, change in dose from initiation, delayed discharge due to uncontrolled pain, opioid related adverse effect, and opioid prescription at discharge.
Results: A total of 138 participants were included in the study. Only 123 were included in the primary outcome after 15 were excluded for the utilization of multiple pain scoring systems. Neither the primary outcome nor any of the secondary outcomes were found to be statistically significant, which suggests that there is no difference in efficacy or safety of parenteral morphine and nalbuphine. Although there was not statistical difference between groups in required dose changes, there was a trend toward an increased frequency in the morphine group.
Conclusions: The data suggests that nalbuphine is as effective as morphine for acute pain management in an inpatient setting when parenteral analgesia is required, with no statistical difference in adverse drug reactions, required dose changes, or in adverse drug reactions. Having an option for treating acute pain that would provide equal pain relief as a long-time, first-line agent while having less frequent and severe side effects could be clinically significant. However, larger, more detailed studies are needed.
********************************************************************************************************************
On Site #42 – Student Research
Title: Dispense this, not that! Requirements for confident opioid dispensing
Submitting Author: Ashley Riley, PharmD Candidate
Authors: Ashley M. Riley, PharmD Candidate, Southern Illinois University Edwardsville; Katrina A. Trentham, PharmD Candidate, St. Louis College of Pharmacy; Nicole M. Gattas, PharmD, BCPS, FAPhA, Associate Professor of Pharmacy Practice, St. Louis College of Pharmacy; Amy M. Tiemeier, PharmD, BCPS, Director, Community Partnerships & Associate Director, Experiential Education & Associate Professor, Pharmacy Practice, St. Louis College of Pharmacy; Halvor T. Olsen, PharmD, Pharmacy Clinical Services Manager, Walmart Health and Wellness; Amy E. Bias, PharmD, Pharmacy Clinical Services Manager, Walmart Health and Wellness; Chris M. Herndon, PharmD, BCACP, Professor, Pharmacy Practice, Southern Illinois University Edwardsville School of Pharmacy.
Organization: Southern Illinois University Edwardsville School of Pharmacy
Abstract
Purpose: As opioid overdose deaths continue to rise across the United States, healthcare providers are taking action to recognize and combat this crisis. Pharmacists play a key role in opioid stewardship, with prescriber and pharmacist communication essential in satisfying corresponding responsibility. This survey aims to identify key information necessary for pharmacists to confidently and responsibly dispense prescribed opioid medications in accordance with laws, policies, and professional judgement. Once identified, key information could be formatted into a universal form or checklist for use by prescribers and pharmacists. A universal tool would improve interdisciplinary communication, reduce patient care delays, promote safe and effective use of opioids, as well as in overall improved patient outcomes. 2.
Methods: The Dispense this, not that! Requirements for confident opioid dispensing survey is a 16-item survey tool (Qualtrics, Provo, Utah, USA) that was distributed electronically to members of the Illinois Pharmacists Association (IPhA) and Missouri Pharmacy Association (MPA) in late 2018. Participation was voluntary and anonymous. The survey questions were designed to identify key information perceived as necessary for pharmacists to confidently dispense opioid prescriptions, as well as information regarding demographics, work setting, location, and general knowledge. The Institutional Review Boards of both Southern Illinois University Edwardsville and St. Louis College of Pharmacy approved the study.
Results: A total of 274 surveys were submitted from members of the Illinois Pharmacists Association and Missouri Pharmacy Association. The majority of pharmacists (greater than 50% of respondents) reported dispensing opioids without the following information: diagnosis, diagnosis code, prior pharmacologic and nonpharmacologic treatment attempted, patient reported pain severity, past medical history, and previous treatment trials with opioids. Information that was deemed not necessary for dispensing opioids by the majority of respondents was patient family history and patient imaging. The majority of respondents deemed the last fill date of any opioid for the patient and confirmation that the prescription drug monitoring database had been reviewed by the prescriber or prescriber’s agent as information that should be required documentation for all opioid prescriptions.
Conclusions: Our findings suggest that, ideally, pharmacists would require documented information from prescribers that is not currently mandated to be on opioid prescriptions. There needs to be improvements in prescriber and pharmacist communication regarding opioid prescriptions in order to satisfy corresponding responsibility, improve patient safety, and improve pain management.
********************************************************************************************************************
On Site #43 – Student Research – Winner Best Student Poster
Title: A Retrospective, Single Center Study Evaluating Readmission Rates and Medications for Patients with Heart Failure with Preserved Ejection Fraction
Submitting Author: Troy Kramer, PharmD Candidate
Authors: Troy Kramer, PharmD candidate; Carrie Vogler, PharmD, Southern Illinois University Edwardsville School of Pharmacy; Mukul Bhattarai, MD, Memorial Medical Center; Robert Robinson, MD, Memorial Medical Center.
Organization: Southern Illinois University Edwardsville
Abstract
Purpose: Heart failure with preserved ejection fraction (HFpEF) has significantly less guideline driven treatment options compared to hearth failure with reduced ejection fraction (HFrEF). This stems from a lack of trials demonstrating medications with improved clinical outcomes for this patient population. The primary objective of this study is to determine which medications and dosages are related to readmission rates for HFpEF patients.
Methods: A retrospective, single center, chart review was performed on patients with HFpEF at an academic medical center. The study was approved by the institution’s IRB. Heart failure patients between the ages of 18-89 with an ejection fraction ≥45% reported on an ECHO were included in the study. Demographic data was also collected. Primary outcomes include 30 day all cause readmission rates along with 30, 60, and 90 day heart failure related readmission rates. Other primary outcomes include prescribing patterns of heart failure medications at discharge and medications prescribed that could be potentially harmful medications in patients with heart failure. Secondary outcomes include patient safety by comparison of readmission rates. Descriptive statistics will be used to analyze the data.
Results: Presented at meeting
Conclusion: Presented at meeting
********************************************************************************************************************
On Site #44 – Student Research
Title: Acute pain management for patients with an opioid dependence disorder receiving Suboxone, naltrexone or methadone
Submitting Author: Erin Lindstrom, PharmD Candidate
Authors: Carrie N. Vogler, PharmD, BCPS, Clinical Associate Professor, Southern Illinois University Edwardsville School of Pharmacy; Katelyn Conklen, PharmD, BCPS, Clinical Pharmacist, Memorial Medical Center; Erin M Lindstrom, PharmD candidate.
Organization: Southern Illinois University Edwardsville School of Pharmacy
Abstract
Purpose: Opioid dependence disorders are treated with medications that alter the opioid receptor response to the offending agents. These actions are desirable for preventing opioid abuse but complicate the treatment for acute pain episodes. The purpose of this study it to determine how acute pain is being treated in this population.
Methods: A retrospective chart review at a non-profit academic medical center was conducted from 3/31/2015 to 3/31/2018 and approved by the institutional review board. Patients admitted to the hospital that are 18-89 years old experiencing acute pain and receiving Suboxone (buprenorphine/naloxone), naltrexone, or methadone for the indication of opioid dependence disorder will be included in this study. Patients on hospice will be excluded. Data will be collected regarding age, gender, reason for hospital admission, significant social history, significant medication history, significant past medical history, opioid dependence therapy, adjuvant therapy used, discharge medications, pain scores and the amount of daily oral morphine milligram equivalents (MME) received. The primary outcomes include type and amount of medication used for inpatient pain management. Safety outcomes include naloxone use, any respiratory rate less than 8 breaths per minute, and daily MME above 50 mg or 90 mg.
Results: Presented at meeting
Conclusion: Presented at meeting
********************************************************************************************************************
On Site #45 – Student Research
Title: Application of the Utah Bleeding Risk Score to LVAD Supported Patients at Advocate Christ Medical Center: A Retrospective Study (Updated title)
Submitting Author: Andrew McInerney, BS, PharmD Candidate
Authors: George Gavrilos, PharmD, MA, Advocate Christ Medical Center; Tatyana Lawrecki, PharmD, BCPS, Advocate Christ Medical Center; Krystina Chickerillo, FNP, ACNS-BC; Nora Krause, FNP, ACNS-BC; Tracy Aicher, FNP, ACNS-BC; Nicole Graney, ACNS-BC; Andrew McInerney, BS, PharmD Candidate, University of Illinois at Chicago College of Pharmacy; Marc McDowell, PharmD, BCPS, Advocate Christ Medical Center; Devin Mehta, MD, Advocate Christ Medical Center; Muhyaldeen Dia, MD, Advocate Christ Medical Center.
Organization: Advocate Christ Medical Center
Abstract
Purpose: Continuous-flow left ventricular assistant devices (CF-LVAD) have improved survival rates in advanced heart failure patients; however, gastrointestinal (GI) bleeding is commonly seen post-implant, resulting in further health complications, increased healthcare expenses, and increased mortality. Recently, researchers at the Utah Transplantation Affiliated Hospitals program developed the Utah Bleeding Risk Score (UBRS) to estimate bleeding risk in this patient population (Yin, et al.). Their study identified seven pre-implant variables most predictive for post-implant GI bleeding. The aim of this study is to evaluate CF-LVAD patients at Advocate Christ Medical Center (ACMC) who have experienced bleeding post-implant and apply the UBRS tool to this patient population.
Methods: CF-LVAD recipients (all devices) at ACMC between 2012 and 2017 who experienced post-implant GI bleeding were evaluated. The following variables were assessed, and patients were assigned one or two points for: age >54 years (1 point), history of previous bleeding (2 points), coronary artery disease (1 point), chronic kidney disease (1 point), severe right ventricular dysfunction (1 point), mean pulmonary artery pressure <18 mm Hg (2 points), and fasting glucose >107 mg/dL (1 point). The risk of each patient having a GI bleed post-implant was then applied using the UBRS, defined as: low (0–1 points), intermediate (2–4), and high risk (5–9). Corresponding 3-year GI bleeding rates found by Yin and colleagues were 4.8%, 39.8%, 83.8% in each risk group respectively.
130 of 562 (23.1%) patients implanted with a CF-LVAD from January 2012 – December 2017 experienced at least one episode of post-implant GI bleeding. Variables assessed are listed below:
Variable |
Results N=130 (%) |
Age >54 |
114 (87.7) |
History of Previous Bleeding |
20 (15.4) |
Coronary Artery Disease |
77 (59.2) |
Chronic Kidney Disease |
31 (23.8) |
Severe Right Ventricular Dysfunction |
3 (2.3) |
Mean Pulmonary Artery Pressure <18 mmHg |
7 (5.4) |
Fasting Glucose > 107 mg/dL |
85 (65.4) |
Utah bleeding scores are listed below:
Risk Score |
Results N=130 (%) |
Low (0–1 Points) |
14 (10.7) |
Intermediate (2–4 Points) |
99 (76.2) |
High (5–9 Points) |
17 (13.1) |
The mean UBRS was 2.83.
Conclusions: Yin and colleagues found the UBRS to be a simple predictive model based on pre-implant clinical factors that can effectively risk stratify CF-LVAD patients based on their probability of GI bleed. In their study, they observed an intermediate risk of GI bleed in all CF-LVAD supported patients based on UBRS. In the subset of CF-LVAD supported patients at ACMC who experienced a post-implant GI bleed, the same intermediate risk probability was observed. Yin’s study found a three-year, 39.8% risk of GI bleed post-implant for intermediate risk patients, and while a direct correlation cannot be made between our two patient populations at this time, we observed a 23.1% overall incidence of post-implant GI bleeding in CF-LVAD supported patients over a five-year period, the majority (76.2%) of whom were found to be at intermediate risk. Prospective, multicenter validation studies should be conducted to further elucidate the accuracy and usefulness of this tool in clinical practice.
********************************************************************************************************************
On Site #46 – Student Research
Title: Rocuronium versus succinylcholine in the traumatically injured brain: A prospective observational cohort (RVSTIB)
Submitting Author: Ammarah Nadeem, PharmD Candidate
Authors: Ammarah Nadeem, PharmD Candidate, Advocate Christ Medical Center; Chuxian Tang, PharmD Candidate, Advocate Christ Medical Center; Samson Tang, PharmD Candidate, Advocate Christ Medical Center; Marc McDowell, PharmD, BCPS, Advocate Christ Medical Center; Sabrin Jaber, PharmD, Advocate Christ Medical Center.
Organization: Advocate Christ Medical Center
Abstract
Purpose: According to the Centers for Disease Control and Prevention, there were approximately 2.8 million traumatic brain injury (TBI)-related emergency department (ED) visits in 2013. Patients with a TBI require rapid sequence intubation (RSI) to prevent hypoxia and death. A recent retrospective study suggests that the use of succinylcholine in patients with severe TBI is associated with increased mortality. However, due to rocuronium’s longer duration of action compared to succinylcholine, this may obscure neurological examination or delay treatment for seizure potentially leading to worse outcomes. This study aims to evaluate the incidence of mortality in patients with a TBI after being administered succinylcholine or rocuronium. (104).
Methods: This is a prospective, single center observational study performed at a large tertiary community teaching hospital in patients greater than 18 years of age presenting with a traumatic brain injury (TBI) requiring rapid sequence intubation (RSI) in the emergency department (ED). Patients who received more than one paralytic or received a paralytic other than rocuronium or succinylcholine for the purpose of RSI, had a surgical airway placed, were< 18 years of age, pregnant, sustained an in-hospital or out-of-hospital cardiac arrest prior to intubation, or had an attempt to intubate outside the ED were excluded. The primary outcome of this study is incidence of mortality. The following data points will be collected: age, sex, race, height, weight, pre-intubation Glasgow Coma Score, baseline serum creatinine and potassium, type of traumatic head injury (defined as blunt or penetrating), mechanism of injury, additional injuries, number of intubation attempts, pre-intubation vitals (oxygen saturation, systolic and diastolic blood pressure, and heart rate), sedative induction agent and dose used for RSI, paralytic agent and dose used for RSI, incidence of mortality, total length of stay (LOS), intensive care unit LOS, type of intracranial injury (defined as: subarachnoid hemorrhage, subdural hematoma, epidural hemorrhage, or intraventricular hemorrhage), presence of mid-line shift, mass effect, or skull fracture, and history of anticoagulant or antiplatelet use. Data will be collected without patient identifiers and analyzed using descriptive statistics. (226).
Results: Out of ten total patients, six were administered succinylcholine and four were administered rocuronium. The incidence of in-hospital mortality was similar in both groups (33% vs 54%, p=0.54). Succinylcholine was associated with a non-significant increase in hospital (6.5 vs 1.8, p=0.10) and ICU LOS (14.8 vs 4.8, p=0.17). (48).
Conclusion: In TBI patients, there is no difference between rocuronium and succinylcholine in the incidence of in-hospital mortality when used for RSI. There was also no difference between groups in ICU or hospital LOS. Larger studies are required in order to meet power and determine significance. (45).
********************************************************************************************************************
On Site #47 – Student Research
Title: Diluted single-syringe administration of adenosine for the rapid conversion of supraventricular tachycardia in the Emergency Department
Submitting Author: Alexandria Nudo, PharmD Candidate
Authors: Alexandria Nudo, PharmD Candidate, Advocate Christ Medical Center; Kevin Johns, PharmD Candidate, Advocate Christ Medical Center; Sonal Sahni, PharmD Candidate, Advocate Christ Medical Center; Robert Mokszyck, PharmD, BCPS, Advocate Christ Medical Center; Neal Lyons, PharmD, BCPS, Advocate Christ Medical Center; Allyson Greenberg, PharmD, BCPS, Advocate Christ Medical Center; Mary Hormese, PharmD, BCPS, Advocate Christ Medical Center; Marc McDowell PharmD, BCPS, Advocate Christ Medical Center.
Organization: Advocate Christ Medical Center
Abstract
Purpose: Supraventricular tachycardias (SVT) are a common cause for emergency department visits. Adenosine is an effective treatment to abate SVT, however differing methods exist for administration. No studies have prospectively evaluated differing methods of administration in a large number of patients. To evaluate the efficacy of adenosine in a single-syringe diluted with normal saline compared to the standard two-syringe method for rapid conversion of SVT to normal sinus rhythm.
Methods: A single center, prospective, observational study was conducted from November 1, 2016 through February 28, 2018 on patients presenting to the emergency department in SVT treated with adenosine. Drug was prepared by the pharmacist and method of administration was at the preference of the physician. Adenosine was either prepared as a single-syringe combined with a 0.9% NaCl flush to a total of 20ml or as the conventional two separate syringes. Rates of conversion from SVT to normal sinus rhythm were recorded.
Results: Presented at meeting
Conclusions: Presented at meeting
College Connection
Chicago State University College of Pharmacy
My First Experience in a Hospital Pharmacy
College Connection
by Makek Hassan - P2, SSHP Member
Midwestern University Chicago Collge of Pharmacy
Guidance for APPE Rotations From a Current 4th Year Pharmacy Student
College Connection
by Dean J. Brock, ICHP member
In order for a student to be successful on each APPE rotation, he or she must be inquisitive, proactive, and professional. Some APPE rotations will be significantly more strenuous than others, but if the student is able to strategize and adapt quickly to the expectations of each individual preceptor, he or she is more likely to be successful. Keeping in mind the sacrifices that were made to get through the didactic portion of pharmacy school, as well as the rewards for those sacrifices after finishing pharmacy school, the motivation to flourish at each APPE rotation should be colossal. ■
Roosevelt University College of Pharmacy
Springing into Something New!
College Connection
by Kristina Khaireddine PS3, SSHP President
Rosalind Franklin University College of Pharmacy
Wellness Week at Rosalind Franklin University of Medicine and Science
College Connection
by Shayda Ashraf, PharmD Candidate 2020, Illinois Council of Health System Pharmacists - Historian
Southern Illinois University Edwardsville School of Pharmacy
My Jamaica Mission: A Life Changing Experience
College Connection
by Paris Smith, P3, Vice President Southern Illinois University Eadwardsville (SIUE) - School of Pharmacy
University of Illinois at Chicago College of Pharmacy
Students Advocating for the Profession of Pharmacy
College Connection
by Shannon Menard, P3
The rest of the week, we set up a booth in the COP lobby to continue the conversation about PAI and promote registration for Legislative Day. Students were invited to fill out a questionnaire asking what provider status for pharmacists would mean to them. This sparked a conversation among the students about how they could advance practice while in pharmacy school as well as when they graduate. Most students discussed how the impact of provider status would impact pharmacy and patient outcomes while others brought up potential issues/obstacles that may be encountered. This lead to a healthy debate on how provider status could be implemented in the state. Our contribution to advocacy also consisted of computer booths to recruit and register students to participate in Legislative Day 2019.

More
Board of Directors
120x120.jpg) Noelle Chapman
President
773-502-0928
E-mail Noelle |
 Travis Hunerdosse
Immediate Past President,
Committee Chair, Nominations Committee
312-926-6124
E-mail Travis
|
 Carrie Vogler
President-Elect
217-545-5394
E-mail Carrie
|
 Kathryn Schultz
Treasurer
312-926-6961
E-mail Kathryn
|
 Ed Rainville
Secretary
309-655-7331
|
 Scott Meyers
Executive Vice President ICHP Office
815-227-9292
E-mail Scott
|
 Amy Boblitt
Regional Director Central
217-788-3015
|
 Elise Wozniak
Regional Director Northern
E-mail Elise
|
 Lynn Fromm
Regional Director Southern
618-391-5539
E-mail Lynn
|
 Mary Lee
Organizational Affairs Director
630-515-7311
E-mail Mary
|
 Karin Terry
Professional Affairs Director
309-655-3390
E-mail Karin
|
 David Martin
Educational Affairs Director
E-mail David
|
 Bernice Man
Marketing Affairs Director
312-694-2878
E-mail Bernice
|
 Christopher Crank
Government Affairs Director
630-978-4853
E-mail Chris
|
 Kristine VanKuiKen
Technician Representative
312-355-2035
E-mail Kristine
|
 Bryan McCarthy
Chairman, New Practitioners Network
773-702-1030
E-mail Bryan
|
 Brian Cryder
Ambulatory Care Network Chair
630-515-7656
E-mail Brian
|
 David Tjhio
Chairman, Committee on Technology
816-885-4649
E-mail David
|
 Jennifer Phillips
Editor & Chairman - KeePosted
630-515-7167
E-mail Jennifer
|
 Milena McLaughlin
Assistant Editor - KeePosted
630-515-7293
E-mail Milena
|
 Sandra Durley
340B Network Chair(312) 996-4940 Email Sandra |
Erin Hermes
Chicago State University College of Pharmacy
|
 Shivek Kashyap
Midwestern University Chicago College of Pharmacy
|
Kristina Khaireddine
Roosevelt University College of Pharmacy
E-mail Kristina |
 Brit Der
Rosalind Franklin University College of Pharmacy
|
 James Reimer
Southern Illinois University Edwardsville School of Pharmacy
|
 Henry Okoroike
University of Illinois at Chicago College of Pharmacy
E-mail Henry
|
 Hannah Dalogdog
University of Illinois at Chicago College of Pharmacy
Rockford Campus
|
Denise Kolanczyk
President
|
Milena McLaughlin
President-elect
|
Erika Hellenbart
Immediate Past President
E-mail Erika
|
David Martin
Treasurer
|
Andrew Merker
Secretary
|
Richard Puccetti
Technician Representative
|
Ed Rainville
President
E-mail Ed
|
Jared Sheley
President
E-mail Jared
|
Billee Samples
President
|
Megan Stoller
President-elect
|
Julie Downen
Immediate Past President
|
President - Rock Valley Society
|
President - Southern IL Society
|
President - Sugar Creek Society
|
ICHP Pharmacy Action Fund (PAC)
As of 4.1.19
(As of 4.1.19)
Welcome New Members!
Aneesh Asokan
Omar Cano
Craig Carrell
Kathi Carrico
Estrella Cervantes
Alicia Davis
Jim Hernandez
Brionna Hudson
Faaieza Khan - Recruited by Erin Hermes
Troy Kramer
Daniella Mazzaro
Michele Monzon-Kenneke
Reem Motan
Savan Patel - Recruited by Faaieza Khan
Sonalie Patel
Timothy Sassack
Dragana Tanasic
Luz Vargas
Kyle Vost
Traci Williams - Recruited by Bryan McCarthy
Gadallah Alawi
Ellen Bennett - Recruited by Richard Puccetti
Refaela Beqi
Kristen Brady
Alexus Brown
Justin Coleman
Beza Daniel
Keah Demmer
Samija Drndar
Marianne Equiban
Shams Fadhil
Eva Galka
Roberto Garcia
Mary Gawron
Amanda Gertz
Danerra Grahn
Keosha Hawkins
Leslie Johnson
Ryan Kelly
Celia Lee
Danielle Lee
Nikola Markoski
Kimberly McCarter
Chul Min
Wasim Mohammad
Makenna Mongan
Michelle Mongan
Jimmy Nguyen
Kyrah Offett
Osayawemwen Osakue
Amanda Page - Recruited by Milena McLaughlin
Ronak Patel - Recruited by Alex Tandyk
Shivani Patel
Heather Powell
Tonya Reid
Adnan Restum
Humna Shaikh
Mohammad Skahkwar
Hasmik Sotelo
Amber Tiffany
Julianne Vanderhoogt-Davis - Recruited by Desi Kotis
Stephane Wabo
Amanda Whistler
Bethany Wond
Megellan Yadao
Dean Brock
Eva Delt
Lori Garrett
Joel Hernandez
Yasmin Malki
Lauren Miskell
John Parise - Recruited by Scott Drabant
Jessica Seadler - Recruited by Beth Cady
Sarah Surmeier
Upcoming Events
Topic: Going down the tubes: drug administration via enteral feeding tubes
(Presentation will be accredited for health-system pharmacists and pharmacy technicians)
May 14, 2019 at 6:00 pm
Blackbird - Chicago, IL
Presentation 1: The Evolution of Quality in the 503B Industry
(Presentation 1 is not available for CPE - sponsored by SCA Pharma)
Presentation 2: 2019 Pharmacy Practice Pearls
(Presentation will be accredited for health-system pharmacists)
May 14, 2019
Topic: The Pharmacists' Patient Care Process (PPCP) and Entrustable Professional Activities (EPAs)
(Presentation will be accredited for health-system pharmacists)
May 23, 2019 at noon
Topic: MERINO trial: Effect of Piperacillin/tazobactam vs Meropenem on 30-day Mortality in Patients with E. Coli or Klebsiella pneumoniae Bloodstream Infection and Ceftriaxone Resistance
See your practice site Champion to participate!
September 12-14, 2019
Drury Lane Conference Center - Oakbrook Terrace, IL
November 15-16, 2019
I Hotel and Conference Center - Champaign, IL
By invitation only
March 27-28, 2020
Embassy Suites Conference Center - East Peoria, IL
◆May 14, 2019 ◆June 11, 2019
◆July 9, 2019 ◆August 13, 2019